The Determination Of Sulphate In Water
As there is no direct measuring Sulphate ISE the analysis is performed using a sample subtraction technique with a Lead ISE as the sensor.
There is no limit to the concentration range of Sulphate being measured. All that is required is that the concentration of Lead Standard used in the method is greater than that of the Sulphate in the sample.
Equipment Required:
- EDT directION QP451 pH/mV meter. (Auto End point and 0.1mV resolution)
- EDT directION 3231 Lead Combination ISE
- EDT directION 21307 1000ppm Lead Standard Solution 500ml
- Glass beaker (250ml), Glass 10 and 100ml Pipettes
- Deionised water in a wash bottle
Method of Analysis:
- Pipette 100ml of the Lead Standard into a clean Glass beaker (250ml)
- Place the EDT directION Lead ISE into the Standard and select mV mode
- Press READ and note the stable mV result. (mV1)
- Pipette 10ml off the Sulphate sample into the standard and stir using the electrode.
- After 1 minute Press READ and note the stable mV result. (mV2)
- Remove the Lead ISE, Rinse thoroughly with deionised water and replace the protective cap
Calculation:
Calculate ΔE by subtracting mV2 from mV1. The unknown Sulphate concentration can be calculated using the following equation:
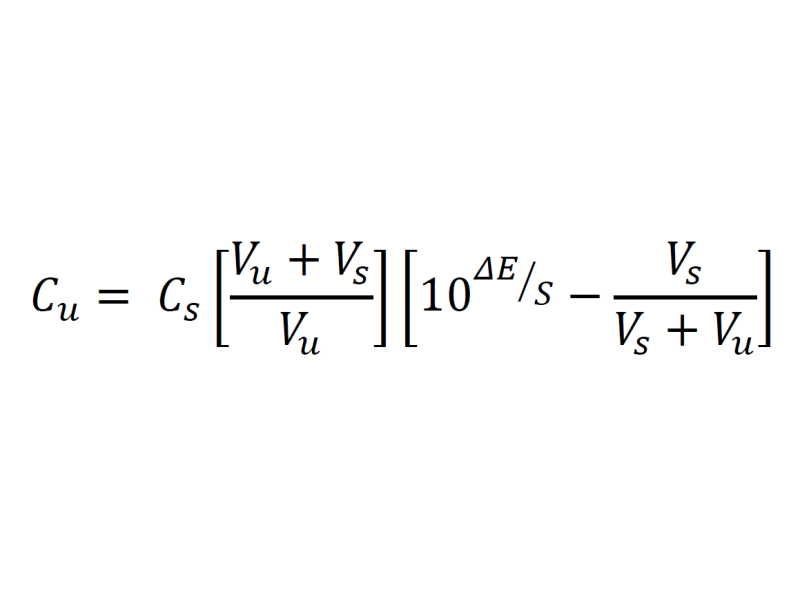
Cu = Concentration of the unknown Sulphate
Cs = Concentration of Pb2+
Vs = Volume of Pb2+ Standard
Vu = Volume of the Sulphate Sample
ΔE = Change in electrode potential in mV
S = Slope of the electrode in mV
Notes: Electrode Slope
To ensure the accurate determination of Sulphate in your sample you will need to measure the slope of the Lead ISE. To do this measure the mV values in 100 and 1000ppm Lead standards. Ideally the standards should contain Lead ISAB Cat no 30307. Subtract the value in 100ppm from the value in 1000ppm. The Slope should be between 24 and 29mV.
If you do not measure the slope we advise you to put 27 into the above formula as this is the most common value for a good Lead ISE.
Visit our YouTube Channel for more content.
